CINA ICH
AI-based triage tool to flag suspected acute Intracranial Hemorrhage on head non-contrast CT Scan
Triage Tool for Detection and Prioritization of traumatic Brain Injuries and Hemorrhagic Stroke on Non-enhanced (non-contrast) head CT images. Five types of ICH are detected: Intraparenchymal, Intraventricular, Subarachnoid, Subdural, and Epidura
Five types of ICH are detected
Intraparenchymal, Intraventricular, Subarachnoid, Subdural, and Epidura
ICH results are sent directly to the PACS
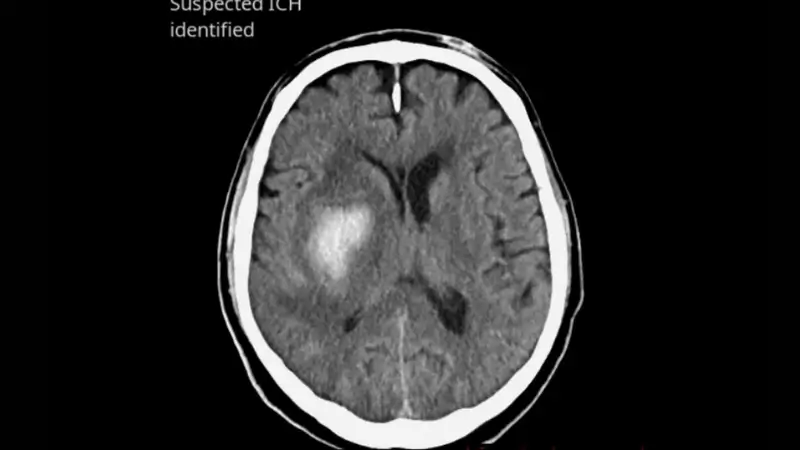
ICH is suspected an axial series is generated, resliced in 5 mm, with a bounding box around the hemorrhage and the mention ‘suspected ICH identified’. If no ICH is identified, a single picture with the mention ‘no findings suggestive of ICH identified’ is sent to PACS
Triage & notification
Information for triage column in the PACS or other notification system can be sent via DICOM or HL7
Reduces turnaround time for head trauma and stroke patients
Helps to quickly and accurately identify ICH to facilitate decision-making, ensure a prompt therapeutic response, and ultimately improve patient outcomes.
Save time, secure your diagnosis and optimize your workflow with Incepto
Publications
-
Soun, J. E., Chow, D. S., Nagamine, M., Takhtawala, R. S., Filippi, C. G., Yu, W., & Chang, P. D. (2021). Artificial Intelligence and
Acute Stroke Imaging. AJNR. American journal of neuroradiology, 42(1), 2–11. https://doi.org/10.3174/ajnr.A6883 -
McLouth, J., Elstrott, S., Chaibi, Y., Quenet, S., Chang, P. D., Chow, D. S., & Soun, J. E. (2021). Validation of a deep learning
tool in the detection of intracranial hemorrhage and large vessel occlusion. Frontiers in Neurology, 12, 656112. https://doi.
org/10.3389/fneur.2021.656112 -
Rava, R. A., Seymour, S. E., LaQue, M. E., Peterson, B. A., Snyder, K. V., Mokin, M., ... & Ionita, C. N. (2021). Assessment of an
artificial intelligence algorithm for detection of intracranial hemorrhage. World Neurosurgery, 150, e209-e217
Regulatory
Contact us to know if this product is available in your country.

